Bi-nuclear Ru(ii) complexes of bis-chalcone and bis-flavonol: synthesis, characterization, photo cleavage of DNA and Topoisomerase I inhibition†
Abstract
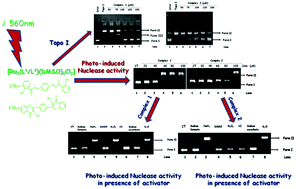
Complexes of type [Ru2(L1/L2)(DMSO)6Cl2] (L1H2 = 1-(2-Hydroxy-phenyl)-3-{4-[3-(2-hydroxy-phenyl)-3-oxo-propenyl]-phenyl}-propenone 1, L2H2 = 1,4-bis-(3-hydroxy-4-oxo-4H-chromen-2-yl)-benzene) 2, are prepared and characterized using spectroscopic techniques. In complex 1, both ruthenium centres possess a similar coordination environment in which all S-dmso coordinate to ruthenium centres. However, in complex 2, one ruthenium centre is coordinated to three S-bonded dmso, and other ruthenium ion is coordinated to two S-bonded dmso and one O-bonded dmso. This difference in coordination around ruthenium centres in complex 2 is supported by two irreversible oxidation peaks observed at 0.84 V and 1.06 V and two MLCT transitions observed at λmax 485 and 565 nm in its cyclic voltammetry and UV-visible spectrum respectively. The interaction of complexes with Calf Thymus DNA (CT-DNA) is monitored using UV–vis titrations (Kb = 2.1 × 106 M−1 for 1 and 1.3 × 105 M−1 for 2) and ethidium bromide displacement studies (Ksv = 1.37 and 0.98). The stronger binding of complex 1 with CT-DNA as compared to complex 2 could be attributed to its conjugated structural framework. In presence of complex 1 under photo-induced condition in visible region at λ 560 nm, supercoiled DNA is converted more in nicked circular form and supported its stronger binding with DNA as compared to complex 2. Complexes intercalate (1 stronger than 2) with DNA. They also prefer minor groove (1) and major groove (2) binding with DNA. Both complexes inhibit topoisomerase I relaxation activity at concentration > 50 μM.

 Please wait while we load your content...
Please wait while we load your content...