Single-phased white-light-emitting KCaGd(PO4)2:Eu2+,Tb3+,Mn2+ phosphors for LED applications†
Abstract
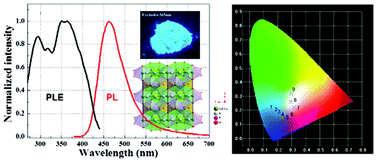
Single-phased white light-emitting KCaGd(PO4)2:Eu2+,Tb3+,Mn2+ phosphors were synthesized by a solid state reaction. By changing the doping contents of Eu2+, Tb3+ and Mn2+, the emission hue could be precisely controlled via the energy transfer mechanism. The structure refinement, luminescence properties as well as their thermal quenching and energy transfer mechanism were firstly investigated. The mechanism of transferring energy between Eu2+, Mn2+ and Tb3+ ions was also discussed in this study. The optimal-composition for white-light is K(Ca0.89Eu0.01Mn0.1)(Gd0.9Tb0.1)(PO4)2, which gives the CIE coordinates of (0.2984, 0.3171). These results indicate that the KCaGd(PO4)2:Eu2+,Mn2+,Tb3+ phosphor could be a promising single-composition phosphor for applications involving white-light NUV LEDs.

 Please wait while we load your content...
Please wait while we load your content...