Effects of bifunctional metal sulfide interlayers on photovoltaic properties of organic–inorganic hybrid solar cells†
Abstract
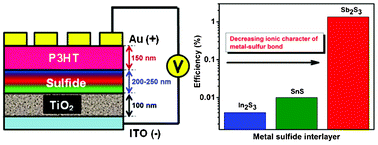
Bilayer organic–inorganic hybrid solar cells have been fabricated with chemical-bath deposited In2S3, SnS, and Sb2S3 interlayers between the organic P3HT hole-conducting and inorganic TiO2 electron-conducting layers. Sb has the highest electronegativity among the elements investigated, and the ionicity of the metal–sulfur bond decreases in the order In2S3 (15%) > SnS (9%) > Sb2S3 (7%). Thus, with a larger covalent character of the Sb–S bond, Sb may bond to S atoms on P3HT relatively easily compared to In or Sn. Such chemical bonding between Sb2S3 and P3HT may then reduce the overall incompatibility between the TiO2 and P3HT layers, making Sb2S3-based devices more efficient compared to In2S3- and SnS-based devices. Along with strong photon absorption, the presence of the metal–sulfur bond promotes improved wetting between the TiO2 sand P3HT layers, resulting in dual functionality for the metal sulfide interlayers. However, along with differences in metal–sulfur bond ionicity, the efficiency improvements seen with Sb2S3 can further be attributed to factors like differences in crystal structure, morphology, photon absorption, charge carrier recombination rates, and the manner in which the sulfide layer interacts with TiO2. While further studies are required to elucidate the exact reason behind the behavior of Sb2S3, the findings discussed herein can provide guidelines for identifying novel absorber layers for developing efficient and cost-effective organic–inorganic hybrid photovoltaics.

 Please wait while we load your content...
Please wait while we load your content...