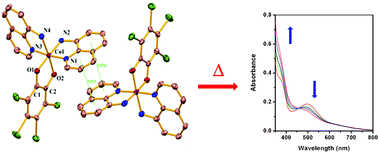
Nine new tetrabromocatechol chelated mononuclear cobalt(III) complexes with various nitrogen-donor ancillary ligands, [Co(en)2(Br4Cat)]ClO4·0·5H2O (1) [Br4CatH2 = tetrabromocatechol, en = ethylenediamine], [Co(pn)2(Br4Cat)]ClO4 (2) [pn = propylenediamine], [Co(dpd)2(Br4Cat)]NO3·CH3OH (3) [dpd = 2,2′-dimethyl-1,3-propanediamine], [Co(amp)2(Br4Cat)]ClO4 (4) [amp = 2-aminomethylpyridine], [Co(aqn)2(Br4Cat)]NO3·CH3OH (5) [aqn = 8-aminoquinoline], [Co(phen)2(Br4Cat)]ClO4·0·5CH3OH (6) [phen = phenanthroline], [Co(bpy)2(Br4Cat)]NO3·Br4CatH2·2CH3OH (7) [bpy = 2,2′-bipyridine], [Et3NH][Co(pym)2(Br4Cat)2]·H2O (8) [pym = pyrimidine] and [Co(tren)(Br4Cat)]NO3·CH3OH·H2O (9) [tren = tris(2-aminoethyl)amine], are reported. All the complexes were characterised by various spectroscopic techniques and electrochemical methods, and their structures have been elucidated by X-ray crystallography. Structural analyses reveal that a 1 : 1 cobalt-dioxolene adduct is the exclusive product with bi- or tetradentate ancillary ligands, while for the monodentate pyrimidine ligand 2 : 1 cobalt-dioxolene is the only outcome. Importantly, an unusual co-crystallization of complex 7 with one molecule of tetrabromocatechol was observed in the crystal lattice. The electrochemical studies of all the complexes disclose quasi-reversible redox processes due to the o-dioxolene ligands, in addition to reductive responses corresponding to the reduction of the cobalt(III) centres to cobalt(II). Although in the solid state all the complexes are diamagnetic, complexes 5 and 8 undergo thermally induced valence tautomerism in solution associated with the drastic change in the optical absorption properties of the complexes in the UV-vis spectroscopy.

 Please wait while we load your content...
Please wait while we load your content...