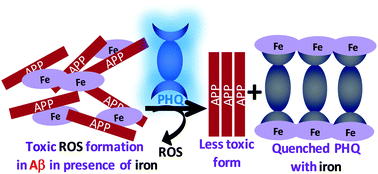
Enhanced levels of toxic metals, especially iron, from the labile iron pool in the brain are primarily responsible for the pathogenesis of several neurological disorders, such as Alzheimer's disease (AD). These metals are a major source for generating highly toxic reactive oxygen species (ROS), accelerating amyloid β (Aβ) peptide aggregation in the brains of AD patients. Aβ has high affinity for iron, resulting in its accumulation and localization in brain plaques enhancing neurotoxic H2O2, oxidative stress and free radical formation. Hence, controlling neurotoxicity would also involve regulation of the redox-active metals present, along with the Aβ. Non toxic conjugated polymer (CP) poly(1,4-bis-(8-(8-hydroxyquinoline)-octyloxy)-benzene) (PHQ) binds iron containing heme and non-heme proteins, such as ferritin, at nanomolar levels with the highest known selectivity (a Stern–Volmer constant (Ksv) value of 0.84 × 107 M−1) in cerebrospinal fluid (CSF) and has been utilized to interact with the bound iron, including non-heme ferritin, in the Aβ protofibril aggregates and diminish their accumulation. The anti-AD activity of PHQ was confirmed via in vitro control studies by doping CSF of healthy individuals (H-CSF) with Aβ(1–40) with and without iron using a Thioflavin-T (ThT) binding assay test and electron microscopy analysis. This conceptually new strategy to clear the cerebral deposits using a CP allows the toxic aggregated Aβ peptide fibrils present in the CSF to be successfully disrupted under physiological conditions.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...