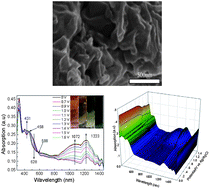
In this work, we synthesized a conjugated polyschiff base (PSB) from N,N′-bis(4-aminophenyl)-N,N′-bis(β-naphthyl)-bisphenylenediamine and 2,5-thiophene-dialdehyde through condensation polymerization, in which the triphenylamine (TPA) core acted as an electron donor and hole transporting material, subsequently the polymer was directly used to react with graphene in the presence of an excess of N-methylglycine to produce a highly soluble RGO covalently functionalized with PSB. The composite was fully characterized by Fourier transform infrared spectroscopy (FTIR), Raman spectrometry, thermogravimetric analysis (TGA), photoluminescence (PL) spectroscopy, UV-Vis absorption spectroscopy, cyclic voltammetry (CV), scanning electron microscopy (SEM), transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS), and atomic force microscopy (AFM). A stable, highly efficient charge-transfer configuration was disclosed and confirmed. The work function values of the PSB and RGO–PSB were obtained by Kelvin probe force microscopy (KPFM) measurements. The devices based on RGO–PSB also showed nonvolatile resistive switching behavior. This composite exhibited higher electrochemical catalytic activity for trinitrotoluene (TNT) and showed excellent electrochromic, acidochromic, and photochromic properties.

 Please wait while we load your content...
Please wait while we load your content...