A study was conducted to survey the tolerance of ring-opening metathesis polymerization (ROMP) with respect to amino acid (a.a.) identity of pentapeptide-modified norbornene-based monomers. A library of norbornyl-pentapeptides was prepared with the general structure, norbornyl-GX2PLX5, where residue ‘X’ was changed at each of the two positions (2 or 5) alternately to consist of the natural amino acids F, A, V, R, S, K, N, T, M, Q, H, W, C, Y, E, Q, and D. Each peptide monomer, free of protecting groups, was mixed in turn under a standard set of polymerization conditions with the ROMP initiator (IMesH2)(C5H5N)2(Cl)2Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHPh. Two sets of polymerization reactions were performed, one with monomer : initiator (M : I) ratio of 20 : 1, and another with M : I of 200 : 1. For the nucleophilic amino acids cysteine and lysine, polymerization reactions were quantitatively compared to those of their protected analogues. Furthermore, we describe polymerization of macromonomers containing up to 30 a.a. to test for tolerance of ROMP to peptide molecular weight. These reactions were studied via SEC-MALS and NMR. Finally, with knowledge of sequence scope in hand, we prepared a set of enzyme–substrate containing brush polymers and studied them with respect to their bioactivity.
CHPh. Two sets of polymerization reactions were performed, one with monomer : initiator (M : I) ratio of 20 : 1, and another with M : I of 200 : 1. For the nucleophilic amino acids cysteine and lysine, polymerization reactions were quantitatively compared to those of their protected analogues. Furthermore, we describe polymerization of macromonomers containing up to 30 a.a. to test for tolerance of ROMP to peptide molecular weight. These reactions were studied via SEC-MALS and NMR. Finally, with knowledge of sequence scope in hand, we prepared a set of enzyme–substrate containing brush polymers and studied them with respect to their bioactivity.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHPh. Two sets of polymerization reactions were performed, one with monomer : initiator (M : I) ratio of 20 : 1, and another with M : I of 200 : 1. For the nucleophilic
CHPh. Two sets of polymerization reactions were performed, one with monomer : initiator (M : I) ratio of 20 : 1, and another with M : I of 200 : 1. For the nucleophilic 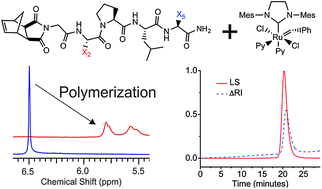

 Please wait while we load your content...
Please wait while we load your content...