Self-healing polymers based on thermally reversible Diels–Alder chemistry
Abstract
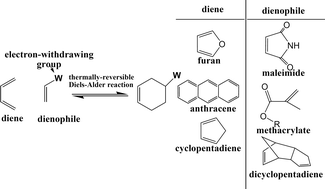
The development of self-healing materials has received much research attention in the last two decades. This review paper gathers recent publications on the self-healing polymeric materials with thermally reversible Diels–Alder (DA) chemistry. The DA reaction is a [4 + 2] cycloaddition involving a diene and a dienophile. The self-healing polymers employing the furan group as a diene and the maleimide group as a dienophile have been widely studied. Multifunctional furan and maleimide compounds construct thermally reversible crosslinked networks showing removability and remendability. Self-healing materials have also been utilized as healing agents for conventional thermosets like epoxy resins. Other diene–dienophile pairs, such as anthracene–maleimide and cyclopentadiene–dicyclopentadiene, have also been utilized for the development of thermally induced self-healing materials. Photo-induced self-healing polymers and some novel applications based on DA reactions have been discussed in this review. Moreover, self-healing polymer systems based on other thermally triggered and assisted reactions are also discussed. The discussed publication has provided promising molecular designs and synthetic strategies for the development of high performance self-healing polymers.

 Please wait while we load your content...
Please wait while we load your content...