Role of graphene on the surface chemical reactions of BiPO4–rGO with low OH-related defects†
Abstract
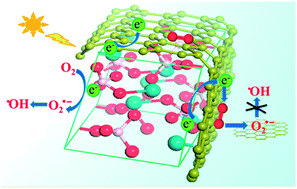
Graphene has been widely introduced into photocatalysis to enhance photocatalytic performance due to its unique physical and chemical properties. However, the effect of graphene on the surface chemical reactions of photocatalysis has not been clearly researched, which is important for photocatalysis because photocatalytic reactions ultimately occur on the catalyst surface. Herein, a two-step solution-phase reaction has been designed to synthesize quasi-core–shell structured BiPO4–rGO cuboids and the role of graphene on the surface chemical reactions was investigated in detail. It was found that the introduced graphene modified the process and the mechanism of the surface chemical reactions. The change mainly originates from the interaction between graphene and the adsorbed O2 molecule. Due to the electron transfer from graphene to adsorbed O2, graphene could tune the interfacial charge transport and efficiently activate molecular oxygen to form O2˙− anions as the major oxidation species instead of ˙OH. In addition, the two-step synthesis approach could efficiently suppress the formation of OH-related defects in the lattice. As a result, the BiPO4–rGO composite exhibited superior photocatalytic activity to BiPO4 and P25, about 4.3 times that of BiPO4 and 6.9 times that of P25.

 Please wait while we load your content...
Please wait while we load your content...