Gold nanoparticles functionalized with a fragment of the neural cell adhesion molecule L1 stimulate L1-mediated functions†
Abstract
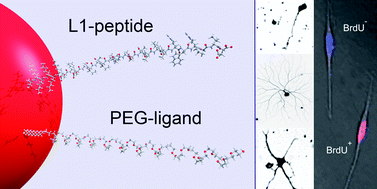
The neural cell adhesion molecule L1 is involved in nervous system development and promotes regeneration in animal models of acute and chronic injury of the adult nervous system. To translate these conducive functions into therapeutic approaches, a 22-mer peptide that encompasses a minimal and functional L1 sequence of the third fibronectin type III domain of murine L1 was identified and conjugated to gold nanoparticles (AuNPs) to obtain constructs that interact homophilically with the extracellular domain of L1 and trigger the cognate beneficial L1-mediated functions. Covalent conjugation was achieved by reacting mixtures of two cysteine-terminated forms of this L1 peptide and thiolated poly(ethylene) glycol (PEG) ligands (∼2.1 kDa) with citrate stabilized AuNPs of two different sizes (∼14 and 40 nm in diameter). By varying the ratio of the L1 peptide–PEG mixtures, an optimized layer composition was achieved that resulted in the expected homophilic interaction of the AuNPs. These AuNPs were stable as tested over a time period of 30 days in artificial cerebrospinal fluid and interacted with the extracellular domain of L1 on neurons and Schwann cells, as could be shown by using cells from wild-type and L1-deficient mice. In vitro, the L1-derivatized particles promoted neurite outgrowth and survival of neurons from the central and peripheral nervous system and stimulated Schwann cell process formation and proliferation. These observations raise the hope that, in combination with other therapeutic approaches, L1 peptide-functionalized AuNPs may become a useful tool to ameliorate the deficits resulting from acute and chronic injuries of the mammalian nervous system.

 Please wait while we load your content...
Please wait while we load your content...