Binuclear organometallic systems incorporating free cyclopentadiene (C5H6) as a ligand rather than the ubiquitous cyclopentadienyl ligand (C5H5) have never been reported in the literature. However, the hexafluorocyclopentadiene ligand (C5F6) is known to form the stable binuclear carbonyl derivatives (η2,η2-C5F6)Fe2(CO)8 and trans-(η3,η1-C5F6)Co2(CO)7. The related hexafluorocyclopentadiene iron carbonyl systems (C5F6)Fe(CO)n (n = 4, 3, 2) and (C5F6)Fe2(CO)n (n = 8, 7, 6, 5) have now been explored using density functional theory. The hexafluorocyclopentadiene ligand is found to use one or both of its C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bonds to bind either as a dihapto η2-C5F6 monoolefin or a tetrahapto η4-C5F6 diolefin in mononuclear (C5F6)Fe(CO)n structures. In binuclear iron carbonyl structures lacking direct iron–iron bonds, such as the lowest energy trans- and cis-(η2,η2-C5F6)Fe2(CO)8 structures as well as higher energy trans-(η2,η2-C5F6)Fe2(CO)n (n = 7, 6, 5) structures, the hexafluorocyclopentadiene ligand binds as a bis(dihapto) diolefin. However, the lowest energy (C5F6)Fe2(CO)n (n = 7, 6, 5) structures have cis stereochemistries with direct iron–iron bonds of orders ranging from one to three. In such lowest energy structures the hexafluorocyclopentadiene ligand is bonded to the Fe2 unit as a trihapto–monohapto η3,η1-C5F6 ligand.
C double bonds to bind either as a dihapto η2-C5F6 monoolefin or a tetrahapto η4-C5F6 diolefin in mononuclear (C5F6)Fe(CO)n structures. In binuclear iron carbonyl structures lacking direct iron–iron bonds, such as the lowest energy trans- and cis-(η2,η2-C5F6)Fe2(CO)8 structures as well as higher energy trans-(η2,η2-C5F6)Fe2(CO)n (n = 7, 6, 5) structures, the hexafluorocyclopentadiene ligand binds as a bis(dihapto) diolefin. However, the lowest energy (C5F6)Fe2(CO)n (n = 7, 6, 5) structures have cis stereochemistries with direct iron–iron bonds of orders ranging from one to three. In such lowest energy structures the hexafluorocyclopentadiene ligand is bonded to the Fe2 unit as a trihapto–monohapto η3,η1-C5F6 ligand.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bonds to bind either as a dihapto η2-C5F6 monoolefin or a tetrahapto η4-C5F6 diolefin in mononuclear (C5F6)Fe(CO)n structures. In binuclear iron
C double bonds to bind either as a dihapto η2-C5F6 monoolefin or a tetrahapto η4-C5F6 diolefin in mononuclear (C5F6)Fe(CO)n structures. In binuclear iron 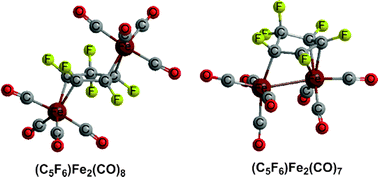

 Please wait while we load your content...
Please wait while we load your content...