The molybdoproteome of Starkeya novella – insights into the diversity and functions of molybdenum containing proteins in response to changing growth conditions
Abstract
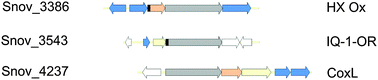
Molybdenum enzymes are known to underpin key reactions in the biological carbon, nitrogen and sulfur cycles, however, the diversity of these enzymes and the reactions they catalyze especially in bacteria is much greater than currently known. We have analysed the molybdoproteome of the soil bacterium Starkeya novella as a function of growth mode and identified a complete pathway for Mo–PPT synthesis, a Mo transporter and 18 gene loci encoding mononuclear Mo-enzymes of the Xanthine Oxidase, Sulfite Oxidase and DMSO Reductase enzyme families. This relatively high number of Mo enzymes may be a specific property of the taxonomic group (Xanthobacteraceae) to which S. novella belongs. About 70% of S. novella Mo enzymes have no characterized close relatives, and two thirds of them are expressed under the conditions analysed, which included heterotrophy, methylotrophy, chemolithotrophy and mixotrophy. Many enzymes were clearly regulated in response to either the type of carbon source present in the growth medium or the presence of thiosulfate, and two, in particular, including an uncharacterized enzyme of the XO family (Snov_3370) were highly abundant under all growth conditions tested. We also uncovered novel enzymes with links to growth in the presence of thiosulfate, such as a PaoABC-type aldehyde oxidoreductase and an uncharacterized group 1 sulfite oxidase family enzyme, although the function of these enzymes during sulfur oxidation is unclear at present. Clearly, further work is needed to uncover the significance of these enzymes for cell metabolism.
- This article is part of the themed collection: Microbial Metallomics

 Please wait while we load your content...
Please wait while we load your content...