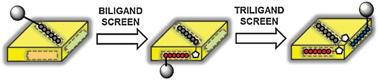
Advances in the fields of proteomics, molecular imaging, and therapeutics are closely linked to the availability of affinity reagents that selectively recognize their biological targets. Here we present a review of Iterative Peptide In Situ Click Chemistry (IPISC), a novel screening technology for designing peptide multiligands with high affinity and specificity. This technology builds upon in situ click chemistry, a kinetic target-guided synthesis approach where the protein target catalyzes the conjugation of two small molecules, typically through the azide–alkyne Huisgen cycloaddition. Integrating this methodology with solid phase peptide libraries enables the assembly of linear and branched peptide multiligands we refer to as Protein Catalyzed Capture Agents (PCC Agents). The resulting structures can be thought of as analogous to the antigen recognition site of antibodies and serve as antibody replacements in biochemical and cell-based applications. In this review, we discuss the recent progress in ligand design through IPISC and related approaches, focusing on the improvements in affinity and specificity as multiligands are assembled by target-catalyzed peptide conjugation. We compare the IPISC process to small molecule in situ click chemistry with particular emphasis on the advantages and technical challenges of constructing antibody-like PCC Agents.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...