Highly effective tandem hydroformylation–acetalization of olefins using a long-life Brønsted acid–Rh bifunctional catalyst in ionic liquid–alcohol systems†
Abstract
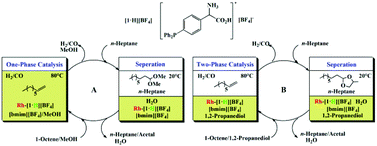
A robust and highly effective tandem hydroformylation–acetalization of olefins using a Brønsted acid–Rh bifunctional catalyst (ARBC) in ionic liquid–alcohol systems is reported. The key feature of the ARBC is its use of a zwitterionic phosphine ligand bearing an amino acid tag. This novel ARBC shows an excellent catalytic efficiency and a long service life without a significant drop in both the hydroformylation efficiency and the acetalization efficiency or Rh loss for more than seventeen cycles. We believe that the long-term high activity and acetal selectivity mainly benefit from the synergy between the acidic active site and the Rh active site on the ARBC and the highly effective immobilization and recycling of ARBC in ionic liquid–alcohol systems due to the strong affinity of ARBC for the ionic liquid.

 Please wait while we load your content...
Please wait while we load your content...