Review of existing experimental approaches for the clinical evaluation of the benefits of plant food supplements on cardiovascular function
Abstract
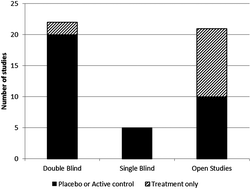
We conducted a survey of the National Centre for Biotechnology Information (NCBI) PubMed database to identify methods most commonly used for the evaluation of the effect of plant food supplements on the cardiovascular system and their relevance to the regulatory status of these products. Particularly, our search strategy was aimed at the selection of studies concerning the clinical evaluation of the beneficial effects of the most commonly studied plant food supplements acting on the cardiovascular system. Following the screening of 3839 papers for inclusion criteria, 48 published reports were retained for this review. Most studies included in this review used a double blind controlled design, and evaluated the effect of plant food supplements on individuals affected by a disease of the cardiovascular system. The majority of the studies were found to be of low methodological quality on the Jadad scale, mainly because of inadequate reporting of adverse events and of patient withdrawals. In comparison, measures used for the evaluation of benefits included mostly cardiovascular risk factors as recommended in international guidelines and in accordance with principles laid down for the evaluation of health claims in food. The risk factors most frequently evaluated belonged to the category of “lipid function and levels”, “heart function” and “blood pressure”. For the absolute majority of the studies, the study period did not exceed one month. This review highlights critical factors to be considered in the design of studies evaluating the health effects of plant food supplements on the cardiovascular system. Between others, the inclusion of healthy individuals, better reporting and description of the characteristics of the product used could improve the quality and relevance of these studies.

 Please wait while we load your content...
Please wait while we load your content...