Post-combustion carbon dioxide capture using electrochemically mediated amine regeneration
Abstract
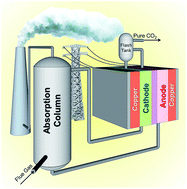
Electrochemically mediated
* Corresponding authors
a
Department of Chemical Engineering, Massachusetts Institute of Technology, 77 Massachusetts Ave., 66-325, Cambridge, MA, USA
E-mail:
tahatton@mit.edu
Tel: +1 617 253-4588
b MIT Energy Initiative, Massachusetts Institute of Technology, Cambridge, MA 02139, USA
Electrochemically mediated

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
M. C. Stern, F. Simeon, H. Herzog and T. A. Hatton, Energy Environ. Sci., 2013, 6, 2505 DOI: 10.1039/C3EE41165F
This article is licensed under a Creative Commons Attribution 3.0 Unported Licence. You can use material from this article in other publications without requesting further permissions from the RSC, provided that the correct acknowledgement is given.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
