Silver enhanced TiO2 thin films: photocatalytic characterization using aqueous solutions of tris(hydroxymethyl)aminomethane
Abstract
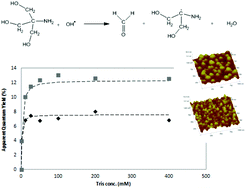
The photocatalytic activity in aqueous solutions of TiO2 and Ag enhanced TiO2 sol–gel produced films was characterized using tris(hydroxymethyl)aminomethane (Tris) under black light (365 nm) and the observed differences in efficiency were further investigated by O2 adsorption studies using the same probe. Hydrogen abstracting species, such as hydroxyl radicals formed upon photocatalysis, are able to abstract hydrogen from Tris. This reaction leads to the formation of formaldehyde which was detected and quantified through a modified version of the Hantzsch reaction. It was found that the Ag enhanced TiO2 film increased the apparent quantum yield from 7% to 12%, partly as a result of a Schottky barrier formation at the metal–semiconductor interface and partly as the sensitizing effect of Ag nanoparticles extends the visible light absorption, which through electron transfer processes enable an efficient charge separation in the TiO2 by attracting acceptor species more efficiently than pure TiO2. The O2 adsorption studies in this paper showed that the Ag enhanced TiO2 film has a stronger adsorption affinity than pure TiO2 towards O2, which make the reduction of O2 more efficient with a subsequent enhanced electron–hole lifetime. It was also found that the Ag enhanced TiO2 film had a poorer adsorption affinity for Tris than the pure TiO2 film, which is a consequence of fewer available surface adsorption sites due to the Ag coverage at 64% which agrees well with the obtained adsorption equilibrium constants (KLH(TiO2) = 615 M−1 and KLH(Ag–TiO2) = 320 M−1).

 Please wait while we load your content...
Please wait while we load your content...