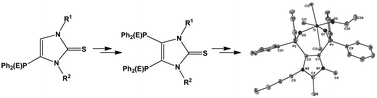
A synthetic route to C4/5-bis(phosphinoyl)imidazole-2-thiones (7d,e) (d: R1 = nBu, R2 = Me; e: R1 = n-dodecyl, R2 = Me) and C4/5-bis(thio/selenophosphinoyl)imidazole-2-thiones (8b,c), (9a,b,e) and 10a (a: R1 = R2 = Me; b: R1 = R2 = Ph, c: R1 = iPr, R2 = Me) is presented that employs initial C5 lithiation of mono-phosphinoyl/thiophosphinoyl substituted imidazole-2-thiones (3c–e)/(4a–c,e) followed by reaction with chlorodiphenylphosphane, leading to mixed phosphinoyl and phosphanyl substituted imidazole-2-thiones (5c–e) or mixed thiophosphinoyl and phosphanyl substituted imidazole-2-thiones (6a–c,e). Subsequent oxidation of mixed phosphinoyl and phosphanyl substituted imidazole-2-thione (5d,e) with H2O2–urea gives the bis(phosphinoyl) substituted imidazole-2-thiones (7d,e), and the oxidation of mixed thiophosphinoyl and phosphanyl substituted imidazole-2-thione (6a–c,e) using H2O2–urea, elemental sulfur or elemental selenium gives a set of mixed P(V)-chalcogenide substituted imidazole-2-thiones (8b,c), (9a,b,e) and 10a, respectively. P(V,V) substituted imidazole-2-thiones 7d and 9a reacted with tellurium tetrachloride, titanium tetrachloride or palladium dichloride to give complexes 11d, (12d and 12d′) and 14a, respectively, having a bidentate chelate (11d and 14a) or a monodentate bonding motif (12d,d′). The titanium complexes 12d,d′ slowly and selectively converted into the mono-ethoxy substituted product 13 possessing a seven membered chelate motif being unprecedented in the titanium chemistry of phosphine oxide donor ligands. The compounds were characterized by elemental analyses, spectroscopic and spectrometric methods and, in addition, X-ray diffraction studies in the case of 5c, 7d, 8b, 9a and 13.

 Please wait while we load your content...
Please wait while we load your content...