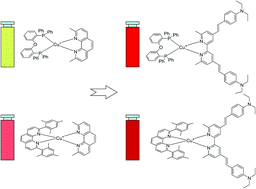
Using the HETPHEN approach, five new heteroleptic copper(I) complexes composed of a push–pull 4,4′-styryl-6,6′-dimethyl-2,2′-bipyridine ligand and a bulky bis[(2-diphenylphosphino)phenyl]-ether (DPEphos) or a bis2,9-mesityl phenanthroline (Mes2Phen) were prepared and characterized by electronic absorption spectroscopy, electrochemistry, and TD-DFT calculations. These complexes exhibit very intense absorption bands in the visible region with extinction coefficient in the range of 5–7 × 104 M−1 cm−1. The analysis of the position, intensity and band shape indicates a strong contribution from an intra-ligand charge-transfer transition centered on the styrylbipyridine ligand along with MLCT transitions. These new complexes experimentally demonstrate that good light harvesting properties with bis-diimine copper(I) complexes are a reality if one chooses suitable ligands in the coordination sphere. This constitutes a milestone towards using bis-diimine copper(I) complexes for solar energy conversion (artificial photosynthesis and solar cells).
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...