Chiral and achiral (imino)phenoxy-based cationic group 4 non-metallocene complexes as catalysts for polymerization of renewable α-methylene-γ-butyrolactones†
Abstract
Protonolysis of M(Bn)4 (M = Zr, Ti; Bn = ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(3,5-tBu2C6H2)OH] in
C(3,5-tBu2C6H2)OH] in ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(3,5-tBu2C6H2)O]Zr(Bn)3 (1) and [(2,6-iPr2C6H3)N
C(3,5-tBu2C6H2)O]Zr(Bn)3 (1) and [(2,6-iPr2C6H3)N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(3,5-tBu2C6H2)O]Ti(Bn)3 (2). A chiral dibenzyl complex 3 incorporating the unsymmetric, tetradentate amino(imino)bis(phenoxy)
C(3,5-tBu2C6H2)O]Ti(Bn)3 (2). A chiral dibenzyl complex 3 incorporating the unsymmetric, tetradentate amino(imino)bis(phenoxy) ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH(2-adamantyl-4-MeC6H2O)]Zr(Bn)2 (3), has also been prepared using the same protonolysis protocol. Abstractive
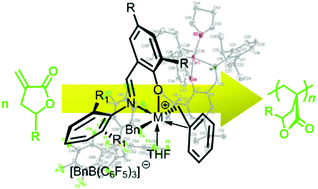
CH(2-adamantyl-4-MeC6H2O)]Zr(Bn)2 (3), has also been prepared using the same protonolysis protocol. Abstractive ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(3,5-tBu2C6H2)O)Zr(Bn)2(THF)]+[BnB(C6F5)3]− (4). Likewise,
C(3,5-tBu2C6H2)O)Zr(Bn)2(THF)]+[BnB(C6F5)3]− (4). Likewise, ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(3,5-tBu2C6H2)O)Ti(Bn)2(THF)]+[BnB(C6F5)3]− (5), accompanied by a small amount of decomposed species as a result of C6F5 transfer. The dibenzyl
C(3,5-tBu2C6H2)O)Ti(Bn)2(THF)]+[BnB(C6F5)3]− (5), accompanied by a small amount of decomposed species as a result of C6F5 transfer. The dibenzyl
- This article is part of the themed collection: Advances in metal-catalysed polymerisation and related transformations

 Please wait while we load your content...
Please wait while we load your content...