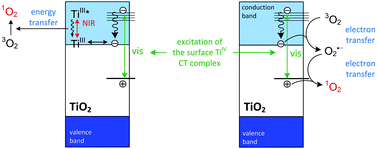
The generation of singlet oxygen in aqueous colloids of nanocrystalline TiO2 (anatase) modified by organic chelating ligands forming surface TiIV complexes was studied. Detailed studies revealed a plausible and to date unappreciated influence of near-infrared irradiation on singlet oxygen generation at the surface of TiO2. To detect 1O2, direct and indirect methods have been applied: a photon counting technique enabling time-resolved measurements of 1O2 phosphorescence, and fluorescence measurements of a product of singlet oxygen interaction with Singlet Oxygen Sensor Green (SOSG). Both methods proved the generation of 1O2. Nanocrystalline TiO2 modified with salicylic acid appeared to be the most efficient photosensitizer among the tested materials. The measured quantum yield reached the value of 0.012 upon irradiation at 355 nm, while unmodified TiO2 colloids appeared to be substantially less efficient generators of singlet oxygen with the corresponding quantum yield of ca. 0.003. A photocatalytic degradation of 4-chlorophenol, proceeding through oxidation by OH˙, was also monitored. The influence of irradiation conditions (UV, vis, NIR or any combination of these spectral ranges) on the generation of both singlet oxygen and hydroxyl radicals has been tested and discussed. Simultaneous irradiation with visible and NIR light did not accelerate OH˙ formation; however, for TiO2 modified with catechol it influenced 1O2 generation. Singlet oxygen is presumably formed according to Nosaka's mechanism comprising O2˙− oxidation with a strong oxidant (hole, an oxidized ligand); however, the energy transfer from NIR-excited titanium(III) centers (trapped electrons) plays also a plausible role.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...