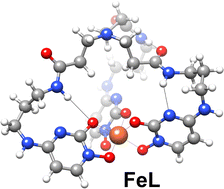
A new tripodal hexadentate ligand, NTP(PrHPM)3, having three hydroxypyrimidinone (HPM) chelating units attached to a nitrilotripropionic acid (NTP) has been prepared and studied in terms of thermodynamic stability of the complexes with iron, aluminium and gallium and it has been subsequently in vivo assayed for its capacity to remove hard metal ions from an animal model overloaded with 67Ga. The anchoring of the HPM units to the NTP scaffold revealed to be an interesting alternative to the reported hexadentate tris(3-hydroxy-4-pyridinone) analogue, NTP(PrHP)3, because the new tris-HPM ligand still keeps high chelating capacity for hard metal ions and presents better water-solubility (log P = −1.51). The in vivo studies show that NTP(PrHPM)3 induces a faster clearance from main organs and an enhancement of overall excretion, as compared with the commercial drug, DFP, or the bidentate HPM compound (HOPY-PrN), albeit slightly lower than the tris-hydroxypyridinone analogue, NTP(PrHP)3. The solution and in vivo results herein presented encourage further studies envisaging the potential clinical applications of hexadentate HPM derivatives as metal sequestering agents.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...