Photocatalytic water splitting under visible light utilizing I3−/I− and IO3−/I− redox mediators by Z-scheme system using surface treated PtOx/WO3 as O2 evolution photocatalyst
Abstract
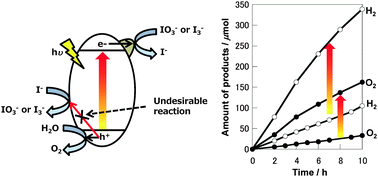
Water oxidation reactions over a surface treated PtOx/WO3 photocatalyst in the presence of IO3− or I3− ions were investigated. When the IO3− ion was used as an electron acceptor, PtOx/WO3 treated by thermal treatment using Cs2CO3 aqueous solution followed by H+-exchange-treatment (PtOx/H-Cs-WO3) showed ca. 4 times higher activity compared with PtOx/WO3 without surface treatment. The apparent quantum yield at 420 nm was 20%. Moreover, when the I3− ion was used as the electron acceptor, the activity of PtOx/WO3 was negligible, while PtOx/H-Cs-WO3 showed a high activity for water oxidation. The cesium surface treatment also had an excellent effect for the improvement of photocatalytic performance of TiO2. The activity of a water splitting reaction under visible light by the Z-scheme system that combined Pt/SrTiO3:Cr/Ta with PtOx/H-Cs-WO3 was 3 times higher than that using PtOx/WO3 as an O2 evolution photocatalyst. Moreover, owing to the contribution from both IO3− and I3− ions as redox mediators, the water splitting reaction stoichiometrically proceeded in a wider pH region (pH 2–9).
- This article is part of the themed collection: Photocatalysis

 Please wait while we load your content...
Please wait while we load your content...