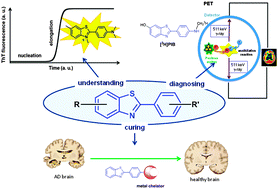
Neurodegenerative disorders including Alzheimer's disease (AD) are drawing scientists' attention within various fields, being one of the most serious diseases mankind will have to fight against in the very near future. AD is multi-factorial and is characterized by two histopathological hallmarks: the senile plaques made of amyloid-β (Aβ) peptide fibrils which also contain high levels of transition metal ions and the neurofibrillary tangles of Tau protein. Aβ aggregation, possibly modulated by metal ions, is now considered as an important factor in AD aetiology. Hence, chemists are studying the details of molecular features at the origin of the disease with special interest in Aβ aggregation and in the design of new molecules for the early diagnosis of AD or with curative properties. In this context, the benzazole molecular scaffold, included for instance in Thioflavin-T, an Aβ fibril specific dye, or the PIB imaging agent, appears to be very attractive and exhibits multiple uses. In the present review, we have thus focused our interest on the applications of benzazole derivatives for understanding, diagnosing and curing AD. After having analysed the synthetic access to 2-arylbenzazoles, we have described a selection of recent applications of such compounds aiming to combat AD. They include the use of Thioflavin-T for the monitoring of Aβ aggregation, the investigations of new PET and SPECT imaging agents for the detection of the senile plaques, the development of bi-functional molecules, encompassing the 2-arylbenzazole moiety for Aβ binding and a chelating unit for metal ions coordination for instance.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...