Catalyst and electrolyte synergy in Li–O2 batteries†
Abstract
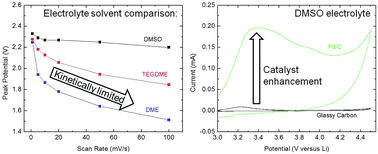
Understanding the interactions between catalyst and electrolyte in Li–O2 systems is crucial to improving capacities, efficiencies, and cycle life. In this study, supported noble metal catalysts Pt/C, Pd/C, and Au/C were paired with popular Li–O2 electrolyte solvents dimethoxyethane (DME), tetraglyme (TEGDME), and dimethyl sulfoxide (DMSO). The effects of these combinations on stability, kinetics, and activity were assessed. We show evidence of a synergistic effect between Pt and Pd catalysts and a DMSO-based electrolyte which enhances the kinetics of oxygen reduction and evolution reactions. DME and TEGDME are more prone to decomposition and less kinetically favorable for oxygen reduction and evolution than DMSO. While the order of oxygen reduction onset potentials with each catalyst was found to be consistent across electrolyte (Pd > Pt > Au), larger overpotentials with DME and TEGDME, and negative shifts in onset after only five cycles favor the stability of a DMSO electrolyte. Full cell cycling experiments confirm that catalyst–DMSO combinations produce up to 9 times higher discharge capacities than the same with TEGDME after 20 cycles (∼707.4 vs. 78.8 mA h g−1 with Pd/C). Ex situ EDS and in situ EIS analyses of resistive species in the cathode suggest that improvements in capacity with DMSO are due to a combination of greater electrolyte conductivity and catalyst synergies. Our findings demonstrate that co-selection of catalyst and electrolyte is necessary to exploit chemical synergies and improve the performance of Li–O2 cells.

 Please wait while we load your content...
Please wait while we load your content...