Rational design, characterization and catalytic application of metal clusters functionalized with hydrophilic, chiral ligands: a proof of principle study†
Abstract
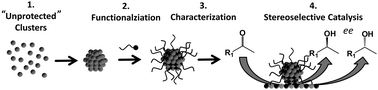
A proof of principle is presented for the rational design of metal clusters functionalized with hydrophilic, chiral ligands. A colloidal method is used to prepare “unprotected” metal clusters of well-defined size that are subsequently functionalized in a separate step with hydrophilic, chiral ligands. As clusters from the same batch are functionalized with different organic molecules while the cluster size is maintained, the approach allows for systematic investigations and the differences in the observed properties to be related to the influence of the functionalizing ligand. Within this work cysteine and two cysteine derivatives (glutathione and N-acetyl-cysteine) are used as functionalizing ligands for Pt clusters. The materials are characterized using various methods allowing for the determination of ligand coverage, binding mode and chiro-optical properties. Finally, 2-butanone hydrogenation is used as a simple model reaction to demonstrate that these systems exhibit the potential to be used as asymmetric, heterogeneous catalysts. The observed differences in selectivity and reactivity are discussed based on the knowledge gained from the characterization.

 Please wait while we load your content...
Please wait while we load your content...