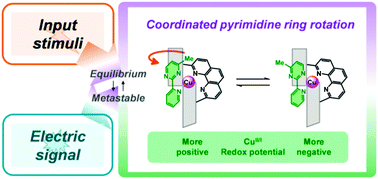
This paper summarizes the results of our recent studies on the development of an artificial molecular rotor system that exhibits a change in redox potential and photoluminescence in response to external stimuli such as heat and photons. The molecular rotor is made of copper complexes bearing two bidentate ligands; the rotor is described here as [Cu(Rpmpy)(Lx)]+, where Rpmpy and Lx are a 4-methyl-2-(2′-pyridyl)pyrimidine derivative and a bidendate ligand with bulky moieties, x, respectively, and the pyrimidine ring can rotate beside the copper centre while maintaining the pyridine–copper connection. The simplicity of the system enabled us to design the rotating motion more accurately. We expected that placing a wall in the rotational trajectory in the Lx moiety would decrease the rate of the rotational dynamics. This slow rate of rotation was a key factor in achieving an external-stimuli-induced switching from thr equilibrium to metastable states. This switching was based on four stable isomers derived from the rotation and oxidation states, the behaviours of which were characterized for isolated copper(I) complexes using spectroscopic and electrochemical measurements at several temperatures. The steric shifts arising from the ring rotation were exploited not only to exhibit well-established oxidation-triggered motion but also to modulate the rest potential of the electrode, to manipulate the intramolecular electron transfer, to develop a redox potential switch based on photo-driven rotation, and to demonstrate the dual-luminescence behaviour.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...