Electrodeposition of germanium from the ionic liquid 1-butyl-1-methylpyrrolidinium dicyanamide†
Abstract
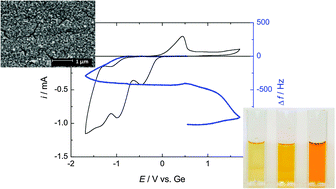
The electrodeposition of germanium from the ionic liquid 1-butyl-1-methylpyrrolidinium dicyanamide ([BMP][DCA]) and a mixture of [BMP][DCA] and 1-butyl-1-methylpyrrolidinium chloride ([BMP]Cl) was studied using cyclic voltammetry and using an electrochemical quartz crystal microbalance (EQCM). [GeCl4(BuIm)2] (BuIm = N-butylimidazole) was used as germanium source as it has a solubility of 0.47 M, up to 13 times the solubility of GeCl4 in [BMP][DCA]. Cyclic voltammograms show an irreversible electrochemical behavior and two reduction waves were observed. The wave at the more positive potential was assigned to the reduction of Ge4+ to Ge2+. The wave at the more negative potential was attributed to the formation of Ge(0). The diffusion coefficient of Ge4+ in [BMP][DCA] containing 0.1 M [GeCl4(BuIm)2] is 1.1 × 10−12 m2 s−1, and the exchange current density is 2 × 10−4 A m−2 at 50 °C. Polymerization of dicyanamide anions took place at the anode in the solution of [BMP][DCA]. The polymerization reaction could be avoided by using an equimolar [BMP]Cl–[BMP][DCA] mixture as electrolyte. Smooth, porous germanium films were electrodeposited on both copper and silicon substrates.

 Please wait while we load your content...
Please wait while we load your content...