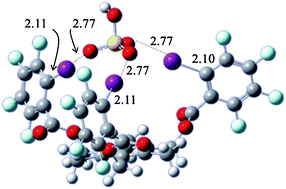
Complexes of mono-, bi- (RB), and tridentate (RT) receptors with a range of anions (Cl−, Br−, I−, NO3−, H2PO4−, HSO4−, and tosylate (TsO−)) have been studied in the gas phase by both experimental and theoretical methods. Temperature dependent blackbody infrared radiative dissociation (BIRD) experiments were performed on complexes of C8F17I with Br− and I−, RB with I−, NO3−, HSO4−, H2PO4−, and TsO−, and RT with I−, HSO4− and TsO− and the observed Arrhenius parameters are reported here. Master equation modeling of the BIRD kinetics data was carried out to determine threshold dissociation energies. Geometry optimizations and thermochemistry calculations were performed using the B3LYP/6-31+G(d,p) level of theory. Additional single point energies were calculated using MP2/6-311++G(2d,p). Results were examined in terms of the binding order of various anions as well as the added binding strength from additional halogen bonding (XB) interactions. The relative binding energies of ions were generally consistent with the ordering previously reported from solution phase experiments; however, the relatively strong binding of H2PO4− to the bidentate receptor contrasted the solution phase observation of oxoanions having weaker interactions when compared to halides. An increase in the energy required to remove the same anion from the tridentate receptor when compared to the bidentate and monodentate receptors is explained as being due to the increase in halogen bonding interactions. The possibility of mixed halogen and hydrogen bonded complexes were considered.

 Please wait while we load your content...
Please wait while we load your content...