Observation of direct vibrational excitation in gas-surface collisions of CO with Au(111): a new model system for surface dynamics
Abstract
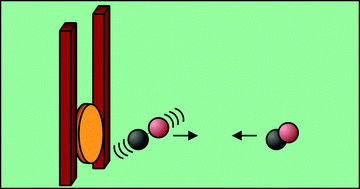
We report vibrational excitation of CO from its ground (v = 0) to first excited (v = 1) vibrational state in collision with Au(111) at an incidence energy of translation of EI = 0.45 eV. Unlike past work, we can exclude an excitation mechanism involving temporary

 Please wait while we load your content...
Please wait while we load your content...