Pseudosymmetry, polymorphism and weak interactions: 4,4′′-difluoro-5′-hydroxy-1,1′:3′,1′′-terphenyl-4′-carboxylic acid and its derivatives†
Abstract
The crystal structures of three derivatives of 4,4′′-difluoro-5′-hydroxy-1,1′:3′,1′′-terphenyl-4′-carboxylic acid are discussed. The acid itself (1), its ethyl ester (2)and hydrazide (3) have been chosen to study the influence of the hydrogen bonding potential on the crystal packing. In 1 and 2 short intramolecular O–H⋯O hydrogen bonds between the hydroxyl and carbonyl groups engage the strong hydrogen bond donors and acceptors, and both these compounds show the effects of packing conflicts. In 1 almost centrosymmetric, stable hydrogen-bonded dimers form between symmetry independent molecules, but the crystal structure is non-centrosymmetric and contains altogether four symmetry-independent molecules (two independent dimers), which show different pseudo-symmetries. In 2 dimer formation is impossible but two different crystal forms of this compound have been found. Both polymorphs crystallize in the P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group and differ mainly in the orientation of the OEt group. In turn in 3 there are no intramolecular hydrogen bonds and the crystal structure is determined mainly by the open motifs created by classical hydrogen bonds and by the complementarity of the respective hydrophilic and hydrophobic parts of the molecule.
space group and differ mainly in the orientation of the OEt group. In turn in 3 there are no intramolecular hydrogen bonds and the crystal structure is determined mainly by the open motifs created by classical hydrogen bonds and by the complementarity of the respective hydrophilic and hydrophobic parts of the molecule.
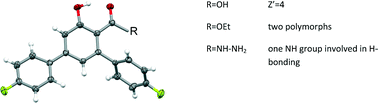

 Please wait while we load your content...
Please wait while we load your content...