Metal–organic gels and coordination networks of pyridine-3,5-bis(1-methyl-benzimidazole-2-yl) and metal halides: self sustainability, mechano, chemical responsiveness and gas and dye sorptions†
Abstract
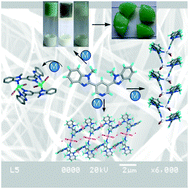
The molecule pyridine-3,5-bis(1-methyl-benzimidazole-2-yl), L1, was shown to form metal–organic gels (MOGs) upon reaction with CuCl2, CuBr2 or CdBr2 in alcoholic solutions. The gels were found to exhibit self sustainability with greater firmness, as suggested by rheological studies. Microscopic analysis, such as POM, SEM and TEM studies, on the MOGs revealed the formation of intertwined gel fibers which provide stability to the gel structures through coordination, hydrogen bonding and π–π interactions. Further crystalline complexes of coordination polymers of L1 with CdBr2, HgCl2, HgBr2, HgI2, CdI2 and CuCl2 were obtained and their single crystal structures have been determined and analyzed in detail. In these structures, the N-atoms of pyridine and benzimidazole were found to coordinate to the metal centers to yield coordination polymers containing one-dimensional helices and one-dimensional chains with M4L4 metallamacrocycles. The structural input from the crystal structures was used to provide probable modes of aggregation in the gels. The MOGs were found to exhibit thixotropic behaviour as well as chemo-responsive behavior. The xerogel materials of the MOGs were found to exhibit an N2 gas sorption property. Furthermore, they were also shown to have potential application for the treatment of waste water containing organic dyes, such as methyl orange and rhodamin B. Furthermore, the obtained results are compared with the non-methylated derivative of L1.
- This article is part of the themed collection: Structural Design of Coordination Polymers

 Please wait while we load your content...
Please wait while we load your content...