M(ii) (M = Mn, Co, Ni) variants of the MIL-53-type structure with pyridine-N-oxide as a co-ligand†
Abstract
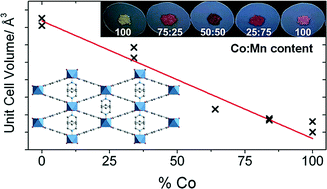
The synthesis and structures of metal–organic framework materials with three-dimensional structures related to the well-known MIL-53 framework are described. These have general chemical formula M(BDC)(PNO), where BDC = 1,4-benzenedicarboxylate, PNO = pyridine-N-oxide, and M = Mn, Co or Ni. The materials contain infinite chains of octahedral metal centres connected by trans corner sharing and cross-linked by 1,4-benzenedicarboxylate anions, where the bridging oxygen atoms in the inorganic chains belong to pyridine-N-oxide ligands. The pyridine rings of these additional ligands project into the extra-framework space, rendering the structure non-porous. The metal ions are present in the +2 oxidation state, as verified using XANES spectroscopy for the case of M = Co. Solid solutions M11−xM2x(BDC)(PNO) can be prepared, which is confirmed using high resolution powder X-ray diffraction. Magnetic susceptibility measurements show antiferromagnetic behaviour at low temperatures for M = Ni or Mn, whereas for M = Co there is evidence for low temperature ferromagnetic interactions. The mean effective magnetic moments deduced per metal ion are consistent with the +2 oxidation states in octahedral coordination.
- This article is part of the themed collection: Structural Design of Coordination Polymers

 Please wait while we load your content...
Please wait while we load your content...