Coordination polymers of a dipyridylazacrown ligand: structural, thermal and spectroscopic properties†
Abstract
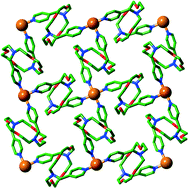
Treatment of the dipyridylazacrown ether ligand N,N′-bis(4-pyridyl-methyl)diaza-18-crown-6 (bpmdc), with hydrated transition metal perchlorate salts resulted in 2D networks with the composition [M(H2O)2(Hbpmdc)2](ClO4)4·xH2O (M2+ = Ni, x = 4 (1); Cu, x = 2 (2) and Zn, x = 4 (3)). The addition of sodium thiocyanate led to displacement of the aqua ligands, yielding [M(NCS)2(Hbpmdc)2](ClO4)2·2H2O (M2+ = Fe (4), Ni (5)), as well as a THF containing variant of Fe (6). A five-coordinate, copper-containing 2D network, [Cu(NCS)(bpmdc)2](NCS) (7), was isolated in the case of copper, while zinc proved to be the exception to the 2D coordination polymer motif, yielding a 1D chain motif [Zn(NCS)2(bpmdc)2] (8). Across these structures the dipyridylazacrown ether ligand showed a remarkably consistent adoption of the S-configuration when empty or singly protonated with a water guest, with bridging lengths varying by only 1.3 Å. The magnetic behaviour of the Fe2+ network 4 was investigated and the d6 compound was found to remain high spin at all temperatures between 300 and 2 K, and thus did not show spin-crossover behaviour.
- This article is part of the themed collection: Structural Design of Coordination Polymers

 Please wait while we load your content...
Please wait while we load your content...