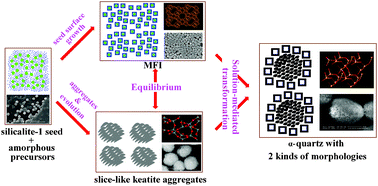
Crystallization–transformation processes in the reaction mixture: (4Na2O–0.1667Al2O3–100SiO2–4000H2O) seeded with 4 wt% of 260 nm silicalite-1 seeds, at 170 °C were investigated by various experimental methods such as, X-ray diffraction (XRD), electron diffraction (ED), scanning-electron microscopy (SEM), transmission electron microscopy (TEM), particle size analysis (PSD), pH measurement, atomic absorption spectrometry (AAS) and dynamic light scattering (DLS). Analysis of the obtained results have shown that the crystallization–transformation takes place by a chain of processes: (i) dissolution of amorphous silica (used as silica source), and formation of 10–50 nm precursor species (PSs), (ii) spending of the PSs for both the limited growth of zeolite ZSM-5 on the surfaces of silicalite-1 seed crystals and formation of worm-like particles (WLPs), (iii) formation of condensed aggregates (CAs) by aggregation/condensation of the WLPs; a part of the composite particles, composed of the silicalite-1 core and ZSM-5 shell are incorporated into CAs, (iv) nucleation of keatite followed by solution-mediated transformation of the condensed WLPs into lamellar keatite, (v) nucleation of α-quartz followed by solution-mediated transformation of keatite into α-quartz; a part of the nuclei of α-quartz are formed on the surface of the core–shell silicalite-1–ZSM-5 composite species and another part of the nuclei are formed in the vicinity of the dissolving keatite and (vi) dissolution of the silicalite-1 core and a major part of the ZSM-5 “interlayer” of the composite particles (α-quartz shell-ZSM-5 “interlayer”-silicalite-1 core) and formation of hollow α-quartz particles with a thin ZSM-5 layer on the inner surfaces of the hollow α-quartz particles. Occurrences of all the mentioned processes are rationally analyzed and explained on the basis of the existing and newly obtained knowledge.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...