Organocatalytic asymmetric desymmetrization: efficient construction of spirocyclic oxindoles bearing a unique all-carbon quaternary stereogenic center via sulfa-Michael addition†
Abstract
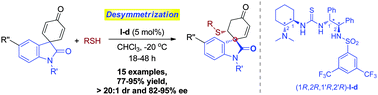
An unprecedented enantioselective desymmetrization of spiro
* Corresponding authors
a
College of Chemistry and Molecular Sciences, Wuhan University, Wuhan 430072, China
E-mail:
cjwang@whu.edu.cn
Fax: +86-27-68754067
b State Key Laboratory of Elemento-Organic Chemistry, Nankai University, Tianjin, China
c School of Pharmaceutical Sciences, Wuhan University, Wuhan 430072, China
An unprecedented enantioselective desymmetrization of spiro

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
L. Yao, K. Liu, H. Tao, G. Qiu, X. Zhou and C. Wang, Chem. Commun., 2013, 49, 6078 DOI: 10.1039/C3CC42587H
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
