Development of poly((3-acrylamidophenyl)boronic acid-co-N,N-methylenebisacrylamide) monolithic capillary for the selective capture of cis-diol biomolecules
Abstract
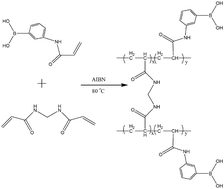
A new hydrophilic boronate affinity monolithic capillary was synthesized for the selective capture of cis-diol biomolecules including glycoproteins and nucleosides, using (3-acrylamidophenyl)boronic acid (APBA) and N,N-methylenebisacrylamide (MBAA) as the functional monomer and cross-linker, respectively. A comparison with other compositionally similar monolithic capillaries was carried out in terms of reversed-phase retention, selectivity, binding capacity and binding pH. The poly(APBA-co-MBAA) monolithic capillary exhibited notably different properties. As both the functional monomer and the cross-linker are hydrophilic, the monolith capillary showed suppressed reversed-phase retention and as a result excellent selectivity toward both nucleosides and glycoproteins was observed. It exhibited the highest binding capacity as compared with other compositionally similar monolithic capillaries. Besides, because of the lower pKa value of APBA, the monolithic capillary was able to selectively capture nucleosides at near-physiological pH.

 Please wait while we load your content...
Please wait while we load your content...