Simple synthesis of yolk-shelled ZnCo2O4 microspheres towards enhancing the electrochemical performance of lithium-ion batteries in conjunction with a sodium carboxymethyl cellulose binder
Abstract
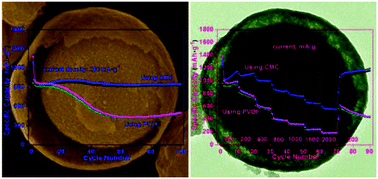
Mixed metal oxides have been attracting more and more attention recently because of their advantages and superiorities, which can improve the electrochemical performance of single metal oxides. These advantages include structural stability, good electronic conductivity, and reversible capacity. In this work, uniform yolk-shelled ZnCo2O4 microspheres were synthesized by pyrolysis of ZnCo-glycolate microsphere precursors which were prepared via a simple refluxing route without any precipitant or surfactant. The formation process of the yolk-shelled microsphere structure during the thermal decomposition of ZnCo-glycolate is discussed, which is mainly based on the heterogeneous contraction caused by non-equilibrium heat treatment. The performances of the as-prepared ZnCo2O4 electrodes using sodium carboxylmethyl cellulose (CMC) and poly-vinylidene fluoride (PVDF) as binders are also compared. Constant current and rate charge–discharge testing results demonstrated that the ZnCo2O4 electrodes using CMC as the binder had better performance than those using PVDF as the binder. It was worth pointing out that the electrode using CMC as the binder nicely yields a discharge capacity of 331 mA h g−1 after 500 cycles at a current density of 1000 mA g−1, which is close to the theoretical value of graphite (371 mA h g−1). Furthermore, the obtained synthetic insights on the complex hollow structures will be of benefit to the design of other anode materials for lithium ion batteries.

 Please wait while we load your content...
Please wait while we load your content...