Unprecedented partial paddlewheel dirhodium methyl isocyanide compounds with unusual structural and electronic properties: a comprehensive experimental and theoretical study†
Abstract
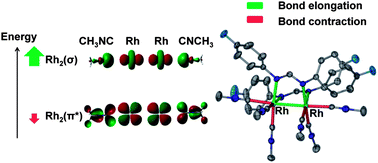
A series of dirhodium acetonitrile compounds, namely cis-[Rh2(DTolF)2(CH3CN)6][BF4]2 (1) cis-[Rh2(F-form)2(CH3CN)6][BF4]2 (2), cis-[Rh2(NNN)2(CH3CN)6][BF4]2 (3) ([F-form]−: p-difluorophenylformamidinate, [NNN]−: p-ditolyltriazenide, [DTolF]−: p-ditolylformamidinate), cis-[Rh2[Ph2P(C6H4)]2(CH3CN)6][BF4]2 (4) and their unprecedented methyl isocyanide analogs 5–8, respectively, were synthesized and characterized by X-ray crystallography, cyclic voltammetry, NMR, electronic and infrared spectroscopies. The elongation of the Rh–Rh distances (∼0.07 Å) and the bonds trans to eq CH3NC in 5–8vs.1–4 are in accord with the strong trans influence of isocyanide. The short Rh–C (CH3NC) distances in 5–8 are attributed to π-backbonding, whereas the short CN distances in CH3NC and the high energy ν(CN) stretches are attributed to appreciable σ-donation of CH3NC. Of particular note are the longer Rh–Rh bonds (∼2.76 Å) that 8 exhibits and unprecedented short ax Rh–C distances in the same range as the eq Rh–C bonds (2.01–2.04 Å) for CH3NC. TD-DFT calculations predict the lowest-energy LMCT transitions in the order 1 < 2 < 5 < 6, 3 < 7 and the HOMO–LUMO energy gaps to be greater in 5–7vs.1–3, respectively, findings corroborated by electrochemical and electronic spectral data. This trend is attributed to the stabilization of the HOMOs and destabilization of the LUMOs in 5–7. The π-backbonding in 5–8 stabilizes the Rh2(π*) orbitals and σ-donation from CH3NC destabilizes the Rh2(σ) orbitals, with the extreme case being 8 where Rh2(σ) becomes the HOMO. This fact accounts for the ∼20-fold increase in intensity of the HOMO Rh2(σ) → LUMO Rh2(σ*) transition for 8 (∼370 nm) vs.4, also in accord with TD-DFT calculations.

 Please wait while we load your content...
Please wait while we load your content...