Theory of multiple proton–electron transfer reactions and its implications for electrocatalysis
Abstract
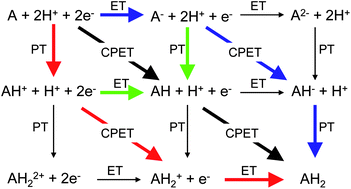
This perspective article outlines a simple but general theoretical analysis for multiple proton–electron transfer reactions, based on the microscopic theory of proton-coupled electron transfer reactions, recent developments in the thermodynamic theory of multi-step electron transfer reactions, and the experimental realization that many multiple proton-coupled electron transfer reactions feature decoupled proton–electron steps in their mechanism. It is shown that decoupling of proton and electron transfer leads to a strong pH dependence of the overall catalytic reaction, implying an optimal pH for high catalytic turnover, and an associated optimal catalyst at the optimal pH. When more than one catalytic intermediate is involved, scaling relationships between intermediates may dictate the optimal catalyst and limit the extent of reversibility that may be achievable for a multiple proton–electron transfer reaction. The theory is discussed in relation to the experimental results for a number of redox reactions that are of importance for sustainable energy conversion, primarily focusing on their pH dependence.

 Please wait while we load your content...
Please wait while we load your content...