Template-free electrosynthesis of gold nanoparticles of controlled size dispersion for the determination of lead at ultratrace levels†
Abstract
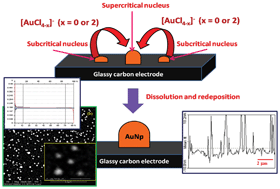
A methodology is presented for the template free electrosynthesis of gold nanoparticles (AuNPs) on a glassy carbon (GC) electrode with controlled particle size dispersion. The mechanism of electrocrystallization at the initial stage was investigated by cyclic voltammetry, chronoamperomentry, chronopotentiometry and in situ spectroelectrochemistry. Two strategies viz. multiple potentiostatic pulse (MPP) and multiple galvanostatic pulse (MGP) were adopted for electrochemically synthesizing the AuNPs. In the MPP strategy, the tapped hemispherical gold nanoparticles (AuNPs(P)/GC) of average diameter of 250–300 nm and average height of 10–15 nm were deposited on a GC electrode covering about 61% of the effective surface area of the electrode. In the MGP strategy, the tapped hemispherical gold nanoparticles (AuNPs(G)/GC) of average diameter of 350–400 nm and average height of 25–30 nm were deposited on a GC electrode covering about 18% of the effective surface area of the electrode. Excellent control of the particle size dispersion was achieved in both the routes of synthesis. The AuNPs, as synthesized, showed excellent electrocatalytic activity despite the absence of any surface stabilizing agent. The synthesized AuNPs showed excellent sensitivity to the determination of Pb(II) by square wave anodic stripping voltammetry (SWASV) by virtue of the underpotential deposition of lead on the gold surface. The limits of detection (LOD) of Pb(II) obtained with a bare GC, bare Au, AuNPs(G)/GC and AuNPs(P)/GC electrodes were calculated as 1.22 μg L−1 (5.86 nM), 122 ng L−1 (587 pM), 86.4 ng L−1 (416 pM) and 57 ng L−1 (274 pM), respectively, at S/N = 3. The analytical response of the AuNPs(P)/GC electrode was found to be the best among the four electrodes. The stability, repeatability, reproducibility and accuracy of the analytical response were found to be satisfactory for the analytical purposes. Bi(III), Cd(II) and Tl(I) showed major interferences with the analytical signal of Pb(II). The concentrations of Pb(II) in the laboratory tap, lake and river water were found to be 1.7 (±2.3%) μg L−1, 3.2 (±1.1%) μg L−1 and 8.4 (±3.9%) μg L−1, respectively using the AuNPs(P)/GC electrode. The results showed a good agreement with the ICP-MS data for the same samples. The studies showed that the concentration of Pb(II) in laboratory tap water, lake water from the Raja Ramanna Centre for Advanced Technology at Indore and water from near the Aassi ghat of the Ganges River in Varanasi, India were below the threshold limit of 10 μg L−1 as per the provisional guidelines set by WHO in 2011.

 Please wait while we load your content...
Please wait while we load your content...