Synthesis of 2,6-disubstituted pyridin-3-yl C-2′-deoxyribonucleosides through chemoselective transformations of bromo-chloropyridine C-nucleosides†
Abstract
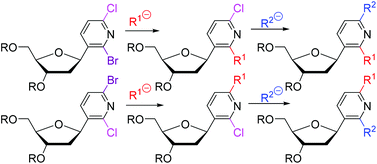
2-Bromo-6-chloro- and 6-bromo-2-chloropyridin-3-yl deoxyribonucleosides were prepared by the Heck coupling of bromo-chloro-iodopyridines with TBS-protected deoxyribose glycal. Some of their Pd-catalyzed cross-coupling reactions proceeded chemoselectively at the position of the bromine, whereas nucleophilic substitutions were unselective and gave mixtures of products. The mono-substituted intermediates were used for another coupling or nucleophilic substitution giving rise to a small library of title 2,6-disubstituted pyridine C-deoxyribonucleosides. The title nucleosides did not exert antiviral or cytostatic effects.

 Please wait while we load your content...
Please wait while we load your content...