Intracellular cleavable poly(2-dimethylaminoethyl methacrylate) functionalized mesoporous silica nanoparticles for efficient siRNA delivery in vitro and in vivo†
Abstract
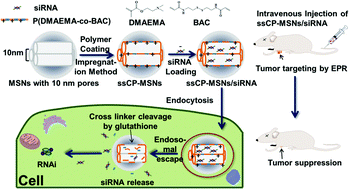
A low cytotoxicity and high efficiency delivery system with the advantages of low cost and facile fabrication is needed for the application of small interfering RNA (siRNA) delivery both in vitro and in vivo. For these prerequisites, cationic polymer–mesoporous silica nanoparticles (ssCP-MSNs) were prepared by surface functionalized mesoporous silica nanoparticles with disulfide bond cross-linked poly(2-dimethylaminoethyl methacrylate) (PDMAEMA). In vitro and in vivo evaluations were performed. The synthesized ssCP-MSNs are 100–150 nm in diameter with a pore size of 10 nm and a positively charged surface with a high zeta potential of 27 mV. Consequently, the ssCP-MSNs showed an excellent binding capacity for siRNA, and an enhancement in the cell uptake and cytosolic availability of siRNA. Furthermore, the intracellular reducing cleavage of the disulfide bonds cross-linking the PDMAEMA segments led to intracellular cleavage of PDMAEMA from ssCP-MSNs, which facilitated the intracellular triggered release of siRNA. Therefore, promoted RNA interference was observed in HeLa-Luc cells, which was equal to that of Lipofectamine 2000. Significantly, compared to Lipofectamine 2000, the ssCP-MSNs were more biocompatible, with low cytotoxicity (even non-cytotoxicity) and promotion of cell proliferation to HeLa-Luc cells. The in vivo systemic distribution studies certified that ssCP-MSNs/siRNA could prolong the duration of siRNA in vivo, and that they accumulated in the adrenal gland, liver, lung, spleen, kidney, heart and thymus after intravenous injection. Encouragingly, with the ability to deliver siRNA to a tumor, ssCP-MSNs/siRNA showed a tumor suppression effect in the HeLa-Luc xenograft murine model after intravenous injection. Therefore, the ssCP-MSNs cationic polymer–mesoporous silica nanoparticles with low cytotoxicity are promising for siRNA delivery.

 Please wait while we load your content...
Please wait while we load your content...