The association of the cytoplasmic domains of interleukin 4 receptor alpha and interleukin 13 receptor alpha 2 regulates interleukin 4 signaling
Abstract
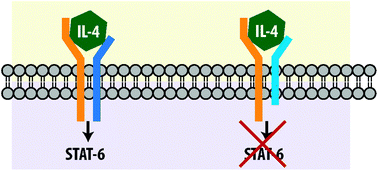
Interleukin-4 (IL-4) and Interleukin-13 (IL-13), key cytokines in the pathogenesis of allergic inflammatory disease, mediate their effects via a receptor composed of IL-13Rα1 and IL-4Rα. A third (decoy) receptor called IL-13Rα2 regulates interleukin signaling through this receptor complex. We employed a variety of biophysical and cell-based techniques to decipher the role of this decoy receptor in mediating IL-4 signaling though the IL-4Rα–IL-13Rα1 receptor complex. Surface plasmon resonance (SPR) analysis showed that IL-13Rα2 does not bind IL-4, and does not affect binding of IL-4 to IL-4Rα. These results indicate that the extracellular domains of IL-4Rα and IL-13Rα2 are not involved in the regulation of IL-4 signaling by IL-13Rα2. We next used a two-hybrid system to show that the cytoplasmic domains of IL-4Rα and IL-13Rα2 interact, and that the secondary structure of the IL-13Rα2 intracellular domain is critical for this interaction. The cellular relevance of this interaction was next investigated. BEAS-2B bronchial epithelial cells that stably express full length IL-13Rα2, or IL-13Rα2 lacking its cytoplasmic domain, were established. Over expression of IL-13Rα2 attenuated IL-4 and IL-13 mediated STAT6 phosphorylation. IL-13Rα2 lacking its cytoplasmic domain continued to attenuate IL-13-mediated signaling, but had no effect on IL-4-mediated STAT6 signaling. Our results suggest that the physical interaction between the cytoplasmic domains of IL-13Rα2 and IL-4Rα regulates IL-4 signaling through the IL-4Rα–IL-13Rα1 receptor complex.

 Please wait while we load your content...
Please wait while we load your content...