Accelerated evaporation of microdroplets at ambient conditions for the on-line analysis of nanoparticles by inductively-coupled plasma mass spectrometry
Abstract
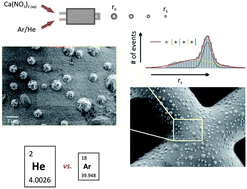
The helium-assisted evaporation of pure, saline, and nanoparticle (NP)-containing microdroplets (∅droplet = 40–50 μm) at ambient conditions was studied. To quantify the liquid-to-gas mass transfer water droplets experience during evaporation, their sizes were monitored in different sections of a custom-made transport system, corresponding to increasing residence times after production. Drying times specified this way turned out to be approximately three times faster than values achievable by heavier gases such as argon which is commonly chosen as the carrier gas for subsequent NP analysis by inductively-coupled plasma mass spectrometry (ICP-MS). Furthermore, residues of saline droplets doped with well-defined amounts of an acidified calcium standard solution were classified by light scattering (LS) and scanning electron microscopy (SEM) indicating nearly complete desolvation over transport distances of a few tens of centimeters if helium or argon–helium mixtures were supplied. Mass transfer rates along droplet trajectories simulated on the basis of computational fluid dynamics (CFD) were found to be consistent with measured ones which proved the applicability of the evaporation model used, thus allowing an optimization of user-defined transport systems without the need for a time-consuming adaptation by trial-and-error. The analytical capabilities of helium-assisted evaporation as an alternative to conventional approaches employing argon-only-based set-ups operated at elevated temperatures were, in addition, demonstrated by ICP-quadruple (Q)MS of gold NPs. Therefore, NP-containing droplets were delivered through an either horizontally or vertically arranged transport assembly both resulting in over-all throughputs of 90 to 100%.
- This article is part of the themed collection: 5 years of nanomaterial analysis by means of ICP-MS

 Please wait while we load your content...
Please wait while we load your content...