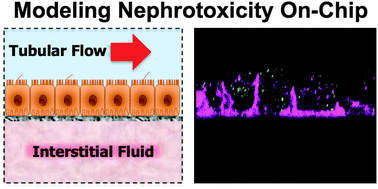
Kidney toxicity is one of the most frequent adverse events reported during drug development. The lack of accurate predictive cell culture models and the unreliability of animal studies have created a need for better approaches to recapitulate kidney function in vitro. Here, we describe a microfluidic device lined by living human kidney epithelial cells exposed to fluidic flow that mimics key functions of the human kidney proximal tubule. Primary kidney epithelial cells isolated from human proximal tubule are cultured on the upper surface of an extracellular matrix-coated, porous, polyester membrane that splits the main channel of the device into two adjacent channels, thereby creating an apical ‘luminal’ channel and a basal ‘interstitial’ space. Exposure of the epithelial monolayer to an apical fluid shear stress (0.2 dyne cm−2) that mimics that found in living kidney tubules results in enhanced epithelial cell polarization and primary cilia formation compared to traditional Transwell culture systems. The cells also exhibited significantly greater albumin transport, glucose reabsorption, and brush border alkaline phosphatase activity. Importantly, cisplatin toxicity and Pgp efflux transporter activity measured on-chip more closely mimic the in vivo responses than results obtained with cells maintained under conventional culture conditions. While past studies have analyzed kidney tubular cells cultured under flow conditions in vitro, this is the first report of a toxicity study using primary human kidney proximal tubular epithelial cells in a microfluidic ‘organ-on-a-chip’ microdevice. The in vivo-like pathophysiology observed in this system suggests that it might serve as a useful tool for evaluating human-relevant renal toxicity in preclinical safety studies.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...