A novel VIVO–pyrimidinone complex: synthesis, solution speciation and human serum protein binding†
Abstract
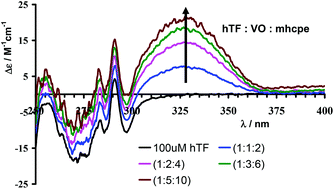
The pyrimidinones mhcpe, 2-methyl-3H-5-hydroxy-6-carboxy-4-pyrimidinone ethyl ester (mhcpe, 1), 2,3-dimethyl-5-benzyloxy-6-carboxy-4-pyrimidinone ethyl ester (dbcpe, 2) and N-methyl-2,3-dimethyl-5-hydroxy-6-carboxyamido-4-pyrimidinone (N-MeHOPY, 3), are synthesized and their structures determined by single crystal X-ray diffraction. The acid–base properties of 1 are studied by potentiometric and spectrophotometric methods, the pKa values being 1.14 and 6.35. DFT calculations were carried out to determine the most stable structure for each of the H2L+, HL and L− forms (HL = mhcpe) and assign the groups involved in the protonation–deprotonation processes. The mhcpe− ligand forms stable complexes with VIVO2+ in the pH range 2 to 10, and potentiometry, EPR and UV-Vis techniques are used to identify and characterize the VIVO–mhcpe species formed. The results are consistent with the formation of VIVO, (VIVO)L, (VIVO)L2, (VIVO)2L2H−2, (VIVO)L2H−1, (VIVO)2L2H−3, (VIVO)LH−2 species and VIVO-hydrolysis products. Calculations indicate that the global binding ability of mhcpe towards VIVO2+ is similar to that of maltol (Hmaltol = 3-hydroxy-2-methyl-4H-pyran-4-one) and lower than that of 1,2-dimethyl-3-hydroxy-4-pyridinone (Hdhp). The interaction of VIVO-complexes with human plasma proteins (transferrin and albumin) is studied by circular dichroism (CD), EPR and 51V NMR spectroscopy. VIVO–mhcpe–protein ternary complexes are formed in both cases. The binding of VIVO2+ to transferrin (hTF) in the presence of mhcpe involves mainly (VIVO)1(hTF)(mhcpe)1, (VIVO)2(hTF)(mhcpe)1 and (VIVO)2(hTF)(mhcpe)2 species, bound at the FeIII binding sites, and the corresponding conditional formation constants are determined. Under the conditions expected to prevail in human blood serum, CD data indicate that the VIVO–mhcpe complexes mainly bind to hTF; the formation of VIVO–hTF–mhcpe complexes occurs in the presence of FeIII as well, distinct EPR signals being clearly obtained for FeIII–hTF and to VIVO–hTF–mhcpe species. Thus this study indicates that transferrin plays the major role in the transport of VIVO–mhcpe complexes under blood plasma conditions in the form of ternary VIV–ligand–protein complexes.
- This article is part of the themed collection: Vanadium in Inorganic Chemistry

 Please wait while we load your content...
Please wait while we load your content...