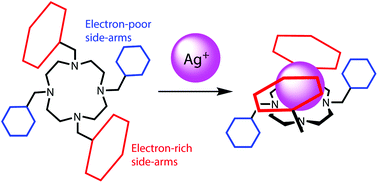
Three tetra-armed cyclens with two kinds of side-arms, 3′,5′-difluorobenzyl/4′-methylbenzyl, 3′,5′-difluorobenzyl/1′-naphthylmethyl, and 3′,5′-difluorobenzyl/9′-anthrylmethyl groups, were prepared by reductive amination of 1,7-bis(3′,5′-difluorobenzyl)-1,4,7,10-tetraazacyclododecane and the corresponding aromatic aldehydes in the presence of NaBH(OAc)3. The X-ray structures of the Ag+ complexes and Ag+-ion-induced 1H NMR spectral changes suggest that (i) the chemical shift changes of the protons at the 2′- and 6′-positions in the 3′,5′-difluorobenzyl/4′-methylbenzyl side-arms are dependent on the electron density on the adjacent substituted benzenes, and (ii) in the tetra-armed cyclens with 3′,5′-difluorobenzyl/1′-naphthylmethyl and 3′,5′-difluorobenzyl/9′-anthrylmethyl groups as side-arms, electron-rich aromatic rings preferentially cover the Ag+ ions incorporated into the ligand cavities, and 3′,5′-difluorobenzyl groups do not participate in the Ag+ interactions. The log K values were estimated using Ag+-ion-induced UV-vis spectral changes.

 Please wait while we load your content...
Please wait while we load your content...