Vapour and mechanically induced chromic behaviour of platinum complexes with a dimer-of-dimer motif and the effects of heterometal ions†
Abstract
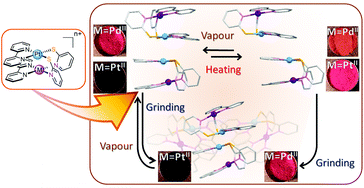
Heterodinuclear complexes, syn-[MPt(μ-pyt)2(bpy)2]n+ (syn-[MPt], M = Pd2+, Au3+, Hpyt = pyridine-2-thiol, bpy = 2,2′-bipyridine), were synthesized as a selective geometrical isomer by stepwise complexation. X-ray analyses of the hexafluorophosphate salts of these complexes proved their dinuclear structures with short M⋯Pt distances (2.9084(4) Å for syn-[PdPt] and 2.9071(4) Å for syn-[AuPt]), similar to the homodinuclear complex (2.9292(2) Å for syn-[PtPt]). In the syn-[PdPt] crystal, two dinuclear motifs are arranged closely in a head-to-head manner with a short Pt⋯Pt distance (3.3757(3) Å), forming a dimer-of-dimer structure as in the case of syn-[PtPt], whereas the corresponding crystal of syn-[AuPt] has a discrete arrangement of the dinuclear motifs. By the isomerisation of syn-[PdPt], anti-[PdPt] with equivalent environments of the Pd2+ and Pt2+ ions was also obtained successfully. Syn-[PdPt](PF6)2 exhibits vapochromic behaviour based on the absorption/desorption of CH3CN vapour, similar to that observed for syn-[PtPt](PF6)2. The reversible structural transformations induced by the uptake and release of CH3CN molecules were investigated by powder and single-crystal X-ray diffraction studies. These revealed that the vapochromic behaviour was based on the interconversion between two phases, the dimer-of-dimer structure with a short Pt⋯Pt distance and a π–π stacked arrangement with no Pt⋯Pt intermolecular interaction. The introduction of the heterometal ions enabled control of the colour region: orange ↔ red for syn-[PdPt] vs. light red ↔ dark red for syn-[PtPt], reflecting the weaker metal–metal interaction between Pd2+ and Pt2+ ions in the dinuclear motif. In addition, these complexes were found to exhibit mechanochromic behaviour based on a crystal-to-amorphous transformation upon grinding, and the reconstruction of the crystal structures by vapour sorption.

 Please wait while we load your content...
Please wait while we load your content...