From well-defined Pt(ii) surface species to the controlled growth of silica supported Pt nanoparticles†
Abstract
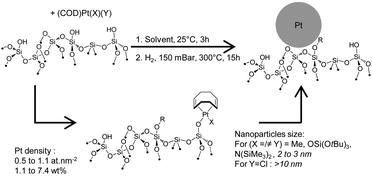
Silica-supported Pt nanoparticles were prepared from well-defined surface platinum(II) surface species, obtained by grafting of well-defined Pt(II) molecular precursors with specific ligands (Cl, Me, N(SiMe3)2, OSi(OtBu)3) onto silica partially dehydroxylated at 200 and 700 °C yielding well-defined platinum(II) surface species. This approach allowed for testing the effect of Pt density and ligands on nanoparticle size. Higher grafting densities are achieved on silica partially dehydroxylated at 200 °C due to its initially higher surface silanol density. Surface species have been synthesized from symmetrical and dissymmetrical complexes, namely (COD)Pt(Me)2, (COD)Pt(OSi(OtBu)3)2, (COD)Pt(Me)(OSi(OtBu)3), (COD)Pt(Me)(N(SiMe3)2), (COD)Pt(Cl)(N(SiMe3)2) and (COD)Pt(N(SiMe3)2)(OSi(OtBu)3) yielding mono-grafted complexes of general formula (COD)Pt(R)(OSi![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) ) according to elemental analyses, diffuse reflectance infrared fourier transform (DRIFT) and carbon-13 solid-state nuclear magnetic resonance (NMR) spectroscopies. While the dimethyl-complex shows low reactivity towards grafting, bis-siloxy and dissymmetric complexes demonstrate better reactivity yielding platinum loadings up to 7.4 wt%. Upon grafting amido complexes, the surface passivation yielding Me3SiOSi
) according to elemental analyses, diffuse reflectance infrared fourier transform (DRIFT) and carbon-13 solid-state nuclear magnetic resonance (NMR) spectroscopies. While the dimethyl-complex shows low reactivity towards grafting, bis-siloxy and dissymmetric complexes demonstrate better reactivity yielding platinum loadings up to 7.4 wt%. Upon grafting amido complexes, the surface passivation yielding Me3SiOSi![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) surface species is demonstrated. Nanoparticles have been synthesized from these well-defined surface species by reduction under H2 at 300 °C, under static or flow conditions. This process yields nanoparticles with sizes ranging from 2 to 3.3 nm and narrow size dispersion from 0.5 to 1.2 nm. Interestingly, the chloride complex yields large nanoparticles from 5 to 40 nm demonstrating the strong influence of chloride over the nanoparticles growth.
surface species is demonstrated. Nanoparticles have been synthesized from these well-defined surface species by reduction under H2 at 300 °C, under static or flow conditions. This process yields nanoparticles with sizes ranging from 2 to 3.3 nm and narrow size dispersion from 0.5 to 1.2 nm. Interestingly, the chloride complex yields large nanoparticles from 5 to 40 nm demonstrating the strong influence of chloride over the nanoparticles growth.

 Please wait while we load your content...
Please wait while we load your content...